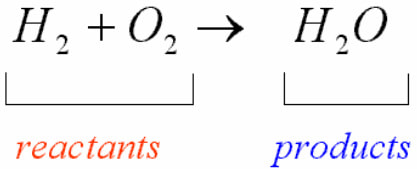Law of conservation of mass
1) Explain the meaning of Law of Conservation of Mass.
Key idea -->> Mass cannot be destroyed or created.
The law of conservation of mass states that mass can neither be created nor destroyed in a chemical reaction. So the amount of matter cannot change.
2) What does it mean by products and reactants?
All chemical reactions involve both reactants and products. Reactants are substances that start a chemical reaction, and products are substances that are produced in the reaction.
1) Explain the meaning of Law of Conservation of Mass.
Key idea -->> Mass cannot be destroyed or created.
The law of conservation of mass states that mass can neither be created nor destroyed in a chemical reaction. So the amount of matter cannot change.
2) What does it mean by products and reactants?
All chemical reactions involve both reactants and products. Reactants are substances that start a chemical reaction, and products are substances that are produced in the reaction.
Word and chemical equations
1. Consider the following word equation:
sodium + chlorine → sodium chloride
a) Is that a chemical reaction? Explain.
Yes, a chemical reaction is a chemical change which forms new substances.
b) What is/are the product(s)?
(The product of the chemical reaction) Sodium Chloride.
c) What is/are the reactant(s)?
(The substances that start the chemical reaction, like the eggs and flour in a cake recipe) Sodium and Chlorine.
2. Consider the following word equation:
sodium + water → sodium hydroxide + hydrogen
a) Is that a chemical reaction? Explain.
Yes, a chemical reaction is a chemical change which forms new substances.
b) What is/are the product(s)?
Sodium Hydroxide and Hydrogen.
c) What is/are the reactant(s)?
Sodium and Water.
3. What are the evidence of a chemical reaction? Give examples.
Evidence of a chemical reaction:
- Change of odor.
- Change of color (for example, silver to reddish-brown when iron rusts).
- Change in temperature or energy, such as the production (exothermic) or loss (endothermic) of heat.
Balancing simple equations
Balance the following equations:
C5H12 + O2 -->CO2 + H2O = C5H12 + 8O2 --> 5CO2 + 6H2O.
Start with C5H12. Because the only thing that has Carbon (C) on the right side of the equation is CO2, then the coefficient for CO2 on the right side -must- be 5.
Then we can look at the H's, in the C5H12. Because ONLY H2O on the right side has H's, the coefficient for the H2O must be 6, in order to get the right side to have the 12 H's we see in the C5H12.
Then, we add together all the O's, we made by changing the coefficients on the right side and we find 5 X 2 = 10 for the CO2 and 6 X 1 = 6 for the H2O.. so we get 16 altogether. Since the only thing with Os on the left side is O2, we can add an 8 coefficient there to get 16.
Zn + HCl --> ZnCl2 + H2 = Zn + 2HCl --> ZnCl2 + H2
https://www.youtube.com/watch?v=pFfm_CbfeqQ
S8 + F2 --> SF6 = S8 + 24F2 --> 8SF6
This involves thinking about multiples!
Let's start with the easier atom to balance.
S8 + F2 → 8SF6
Left side: S = 8; F = 2
Right side: S = (1 x 8) = 8; F = (6 x 8) = 48
Please notice that since SF6 is a substance, you need to multiply the coefficient, not only to S but also to F.
Now to balance the F.
S8 + 24F2 → 8SF6
Left side: S = 8; F = (2 x 24) = 48
Right side: S = (1 x 8) = 8; F = (6 x 8) = 48
C2H6 + O2 --> CO2 + H2O = 2C2H6 + 7O2 --> 4CO2 + 6H2O
https://www.youtube.com/watch?v=XEToG_oCBfA
Acids and Bases
1) Explain the properties of acids and bases. Use a T-chart to help your to explain.
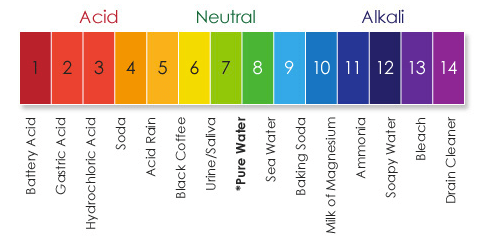
2) Give an example of a strong acid, a weak acid, a strong base and a weak base.
Strong acid - Battery Acid
Weak acid - Soda
Strong base - Drain Cleaner
Weak base - Baking Soda
Strong acid - Battery Acid
Weak acid - Soda
Strong base - Drain Cleaner
Weak base - Baking Soda
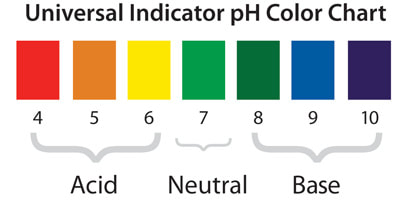
3) If the pH level of a solution is 1, what kind of substance is that?
Acid.
4) What substance can neutralise an acid?
Base.
5) What is the pH of water in general?
****Double check if your teacher wants a range or one value.
The normal range for pH of water systems is 6 to 8.5.
The pH of pure water is typically 7.
6) Which indicator is used to see if the solution is acid or base? How does it work?
****Double check if your teacher wants to know about Universal Indicator or Litmus Paper.
Universal Indicator.
It works like a colour dye, changing the colour of the solution. Then you can use a pH scale to determine if the solution is acidic or basic.
Litmus Paper.
It is a paper coated with an organic dye that changes colour when exposed to an acid or base.
Red litmus paper reacts to basic substances by turning blue, while blue litmus paper reacts to acidic substances by turning red.
Neutralisation
1) Explain the meaning of neutralisation.
The reaction of an acid and a base is called neutralisation. In this reaction, the acidity of an acid is neutralised by a base. At the same time, the alkalinity of the base is neutralised by the acid.
Key idea -->> acid always neutralises a base, and a base always neutralises an acid.
2) Write the word equation of neutralisation.
Acid + Base --> Salt + Water
3) What can neutralise a bee sting and a wasp sting? Why?
1. Wasp sting venom is alkaline (base) and so its effects can be neutralised with vinegar (acid).
2. Bee sting venom is acidic and so its effects can be neutralised with bicarb soda (base).
4) Which substance can neutralise lemon juice? Why?
Baking Soda because it is a base which can neutralise light acids.
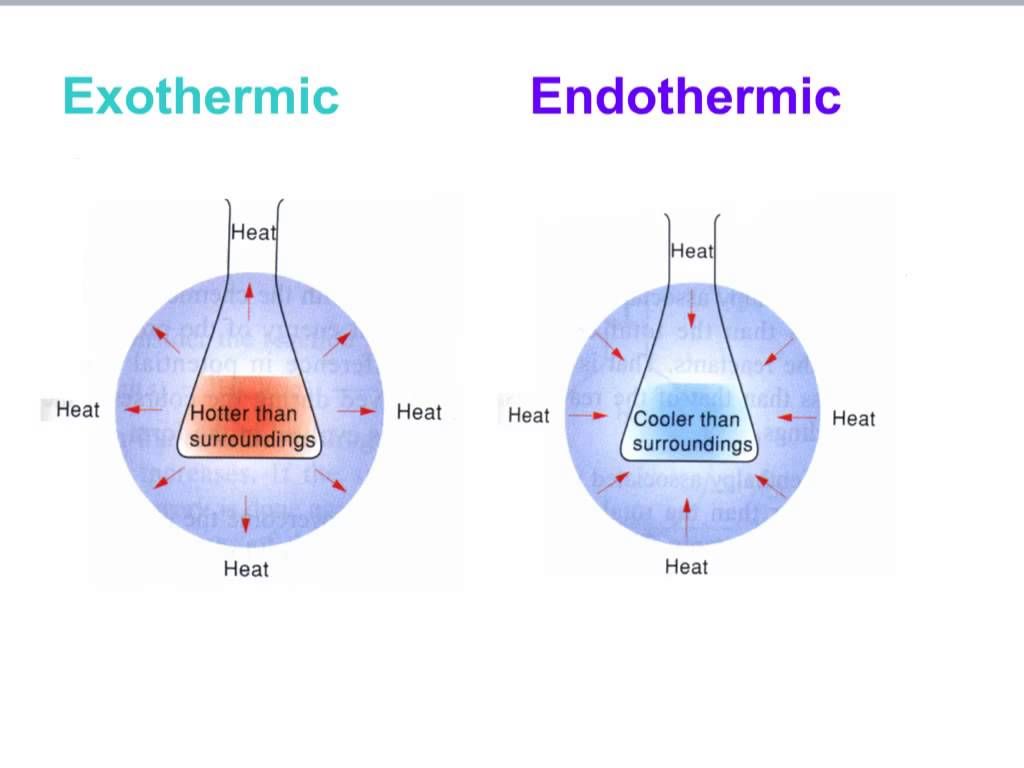
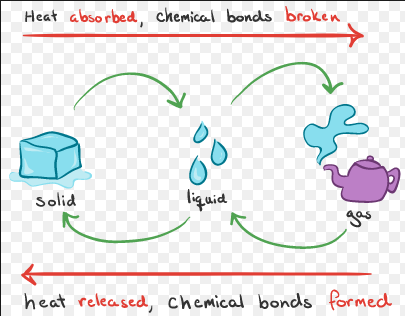
Endothermic and Exothermic reactions
An endothermic reaction takes place when energy is absorbed from surroundings in the form of heat, and exothermic is when energy is released from the system into the surroundings.
1) Explain the meaning of neutralisation.
The reaction of an acid and a base is called neutralisation. In this reaction, the acidity of an acid is neutralised by a base. At the same time, the alkalinity of the base is neutralised by the acid.
Key idea -->> acid always neutralises a base, and a base always neutralises an acid.
2) Write the word equation of neutralisation.
Acid + Base --> Salt + Water
3) What can neutralise a bee sting and a wasp sting? Why?
1. Wasp sting venom is alkaline (base) and so its effects can be neutralised with vinegar (acid).
2. Bee sting venom is acidic and so its effects can be neutralised with bicarb soda (base).
4) Which substance can neutralise lemon juice? Why?
Baking Soda because it is a base which can neutralise light acids.
Endothermic and Exothermic reactions
An endothermic reaction takes place when energy is absorbed from surroundings in the form of heat, and exothermic is when energy is released from the system into the surroundings.
Use a T-Chart to compare the endothermic and exothermic reactions.
Difference |
Endothermic |
Exothermic |
reactions in the environment |
An endothermic reaction takes place when energy is absorbed from surroundings in the form of heat. |
An exothermic reaction is when energy is released from the system into the surroundings. |
reactant |
The endothermic reaction must be supplied with heat - think about the cooking an egg example. The egg needs the heat from the burner/stove to cook. |
The exothermic reaction doesn't need to be supplied with heat. |
reactions in the environment |
An endothermic reaction will cool their surroundings because the reactions draw the heat energy into themselves. |
Exothermic reactions make the surrounding environment hotter since heat energy is released - think about how you would feel warmer sitting in front of a campfire. |
energy |
Endothermic reactions also contain more energy. |
Exothermic reactions will contain less energy. |