Bonds
|
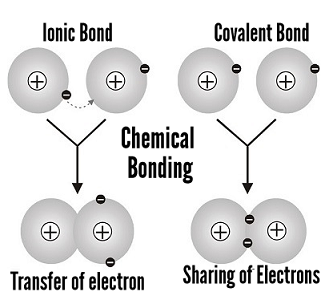
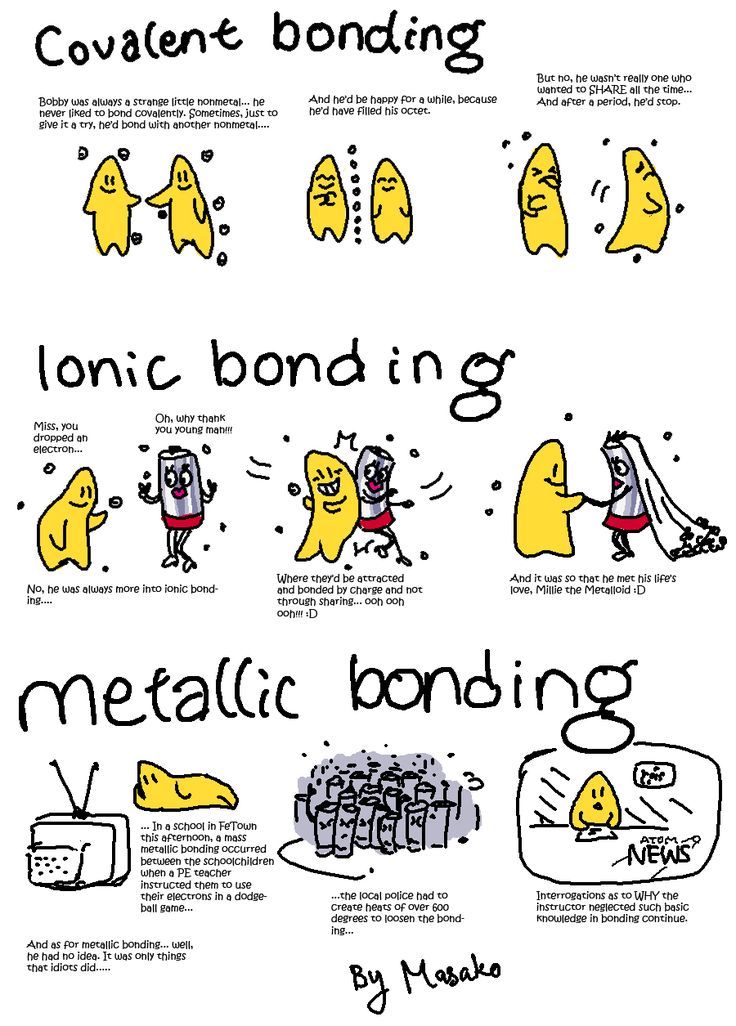
Covalent bond: non-metal and non-metal. Most common bond in organic molecules, this bond involves the sharing of electrons between two atoms.
Ionic bond: metal and non-metaI. Involves a transfer of an electron, so one atom gains an electron while one atom loses an electron. Metallic bonds: the particles in a metal are held together by metallic bonds. Formed from the attraction between mobile electrons and fixed, positively charged metallic atoms. |
Ionic Bond
Ionic bonding is the transfer of valence electrons between a metal and a non-metal.
It is a type of chemical bond that generates two oppositely charged ions. The metal loses electrons to become a positively charged cation, and the non-metal accepts those electrons to become a negatively charged anion.
It is a type of chemical bond that generates two oppositely charged ions. The metal loses electrons to become a positively charged cation, and the non-metal accepts those electrons to become a negatively charged anion.
Covalent Bond
Covalent bonding is the sharing of valence electron pairs between two non-metal atoms.
These electron pairs are known as shared pairs or bonded pairs. The shared pair of electrons holds the two atoms together.
These electron pairs are known as shared pairs or bonded pairs. The shared pair of electrons holds the two atoms together.
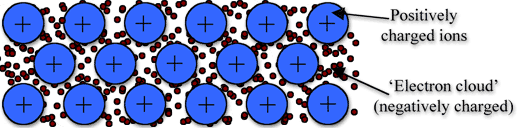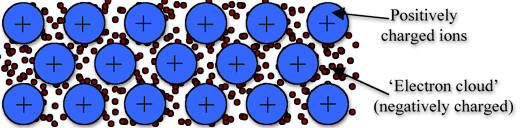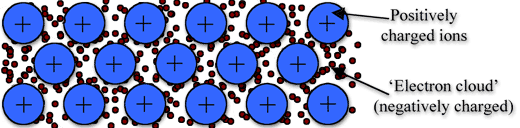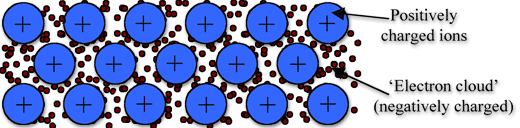
Metallic Bond
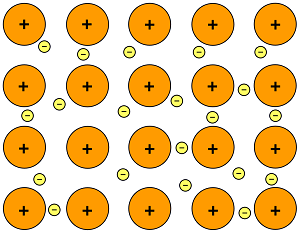
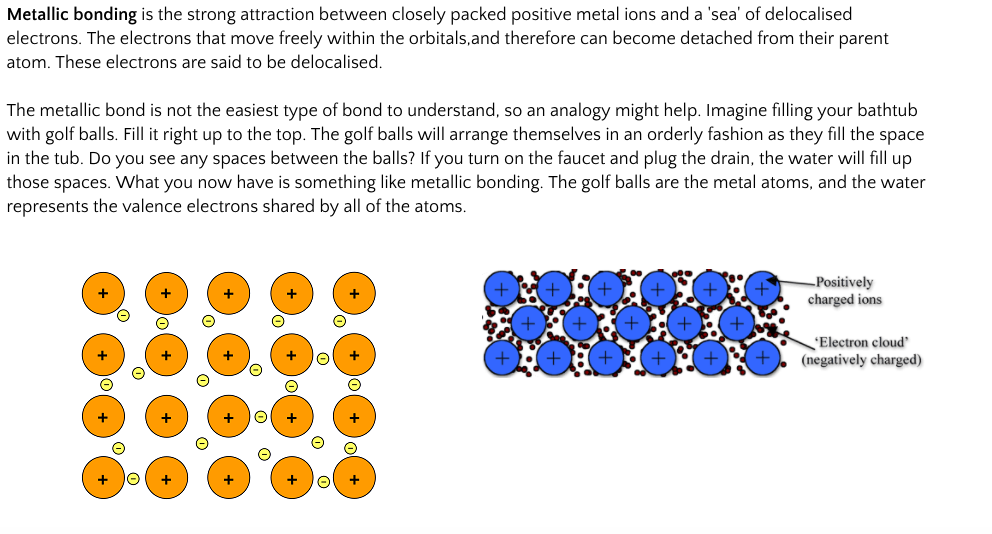
Metallic bonding is the strong attraction between closely packed positive metal ions and a 'sea' of delocalised electrons. The electrons move freely within the orbitals,and therefore can become detached from their parent atom. These electrons are said to be delocalised.
The metallic bond is not the easiest type of bond to understand, so an analogy might help. Imagine filling your bathtub with golf balls. Fill it right up to the top. The golf balls will arrange themselves in an orderly fashion as they fill the space in the tub. Do you see any spaces between the balls? If you turn on the faucet and plug the drain, the water will fill up those spaces. What you now have is something like metallic bonding. The golf balls are the metal atoms, and the water represents the valence electrons shared by all of the atoms.
The metallic bond is not the easiest type of bond to understand, so an analogy might help. Imagine filling your bathtub with golf balls. Fill it right up to the top. The golf balls will arrange themselves in an orderly fashion as they fill the space in the tub. Do you see any spaces between the balls? If you turn on the faucet and plug the drain, the water will fill up those spaces. What you now have is something like metallic bonding. The golf balls are the metal atoms, and the water represents the valence electrons shared by all of the atoms.
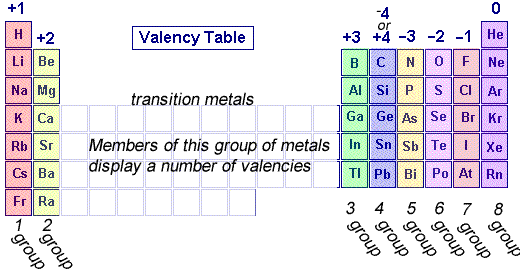
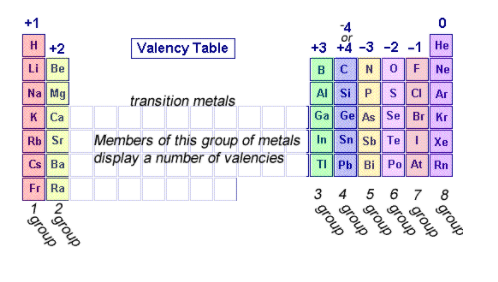
Valence Electrons
1, 2, 3 |
Lose |
Metals |
5, 6 , 7 |
Gain |
Non-Metals |
4 |
Share |
Metalloids |
Writing Ionic Compounds
- We know that positive and negative elements are attracted to each other.
- We know that a compound contains atoms of different elements.
- We must remember that positive element is written first.
- We must remember to change the last syllable to -ide to the end of the negative element.
- E.G: sodium + chlorine -> sodium chloride (NaCl)
- We must swap the valency.